Learn how to set up an ICE Table step-by-step. This guide covers everything from basic stoichiometry with limiting reactants to understanding true chemical equilibrium where reactants never reach zero.
🔗 Reference Video: Watch on YouTube
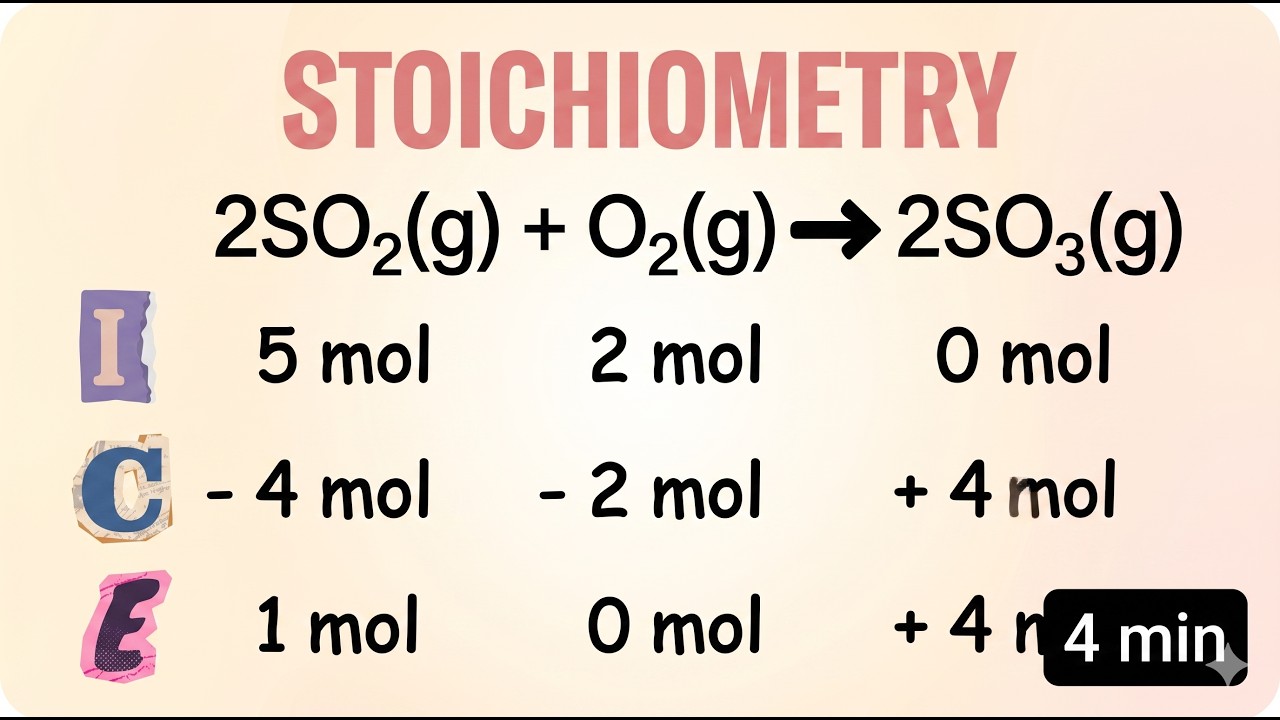
The chemical equation for the formation of SO3 from SO2 and O2 shows a 2:1:2 reaction ratio. Understanding this stoichiometric ratio is the crucial first step in determining exactly how molecules will react.
ICE stands for Initial, Change, and End (or Equilibrium). It is a highly effective, structured method used in chemistry to track the changing amounts of reactants and products throughout a reaction process.
In a reaction that goes 100% to completion, the limiting reactant is completely consumed first. By identifying it, you can accurately calculate the negative and positive changes for all other substances involved.
Unlike a fully completed reaction, a true chemical equilibrium means it is impossible for any reactant to completely disappear. Both reactants and products must coexist, meaning no concentration will ever reach zero.
If we know the final amount of a product (for example, 0.5 moles of SO3), we can work backwards using the 2:1:2 stoichiometric ratio to find the exact equilibrium concentrations of all remaining components.